
This dynamic change in mitochondria can typically be simply characterized by morphological heterogeneity. Mitochondrial dynamics involves continuous fission and fusion, forming a dynamic network to maintain their abundance, morphology, and quality and cell function ( Bereiter-Hahn and Vöth, 1994 Merz et al., 2007). Dynamic changes in mitochondrial morphology are the basis for mitochondrial functionality ( Chen and Chan, 2005 El-Hattab et al., 2018). Mitochondria are highly dynamic organelles that are key players in various biological processes in stem cells, including energy metabolism, oxidative stress reaction, calcium balance, and cell apoptosis. Understanding the etiology of MSC dysfunction under stress and the underlying mechanisms would uncover unique avenues for novel and effective therapeutic strategies in MSC-based regenerative medicine. However, the effectiveness of MSCs is unstable since MSC fate is easily affected by the surrounding microenvironment, involving a complex regulation network ( Sisakhtnezhad et al., 2017), and both endogenous and exogenous MSCs inevitably face harsh surrounding conditions and oxidative stress around the defected tissues ( Chen et al., 2016 Sisakhtnezhad et al., 2017). In recent years, MSCs have emerged as a promising tool for tissue repair and regeneration due to their multilineage differentiation potential, angiogenesis promotion, and immunomodulatory capacity ( Chamberlain et al., 2007 Rajabzadeh et al., 2019). Moreover, MSCs play a key role in the bone marrow microenvironment, which supports and regulates the stem cell niche and hematopoiesis ( Wu et al., 2018). Dysregulation of osteo-adipogenic differentiation balance of MSCs contributes to development of bone diseases, such as osteoporosis, which manifests typically as a lineage shift toward adipocytes instead of osteoblasts in MSCs ( Qi et al., 2017). Increasing evidence suggests that the differentiation of MSCs into adipocytes or osteoblasts is competitively balanced ( Li et al., 2016), and this delicate balance is important for the maintenance of bone homeostasis ( Hu et al., 2018b Muruganandan et al., 2020). In addition to osteoblasts, MSCs are also able to differentiate into adipocytes within bone marrow microenvironment. As the progenitor cells of osteoblasts and osteocytes, MSCs can migrate to defective sites and initiate new bone formation during the early stage of bone healing ( Tang et al., 2009). MSCs are important for preserving tissue homeostasis and have regeneration potential ( Bianco, 2014). Mesenchymal stem cells (MSCs) are multipotent stromal cells that originate from many connective tissues and can differentiate into a variety of cell types, such as osteoblasts, adipocytes, and myoblasts. These findings shed light on the contribution of mitochondrial dynamics to MSC fate and MSC-based tissue repair. We highlight mechanistic insights into modulating mitochondrial dynamics and mitochondrial strategies for stem cell-based regenerative medicine. This review focuses on the role of mitochondrial dynamics in MSC commitment under physiological and stress conditions. The coordination of mitochondrial fission and fusion is crucial for cellular function and stress responses, while abnormal fission and/or fusion causes MSC dysfunction. 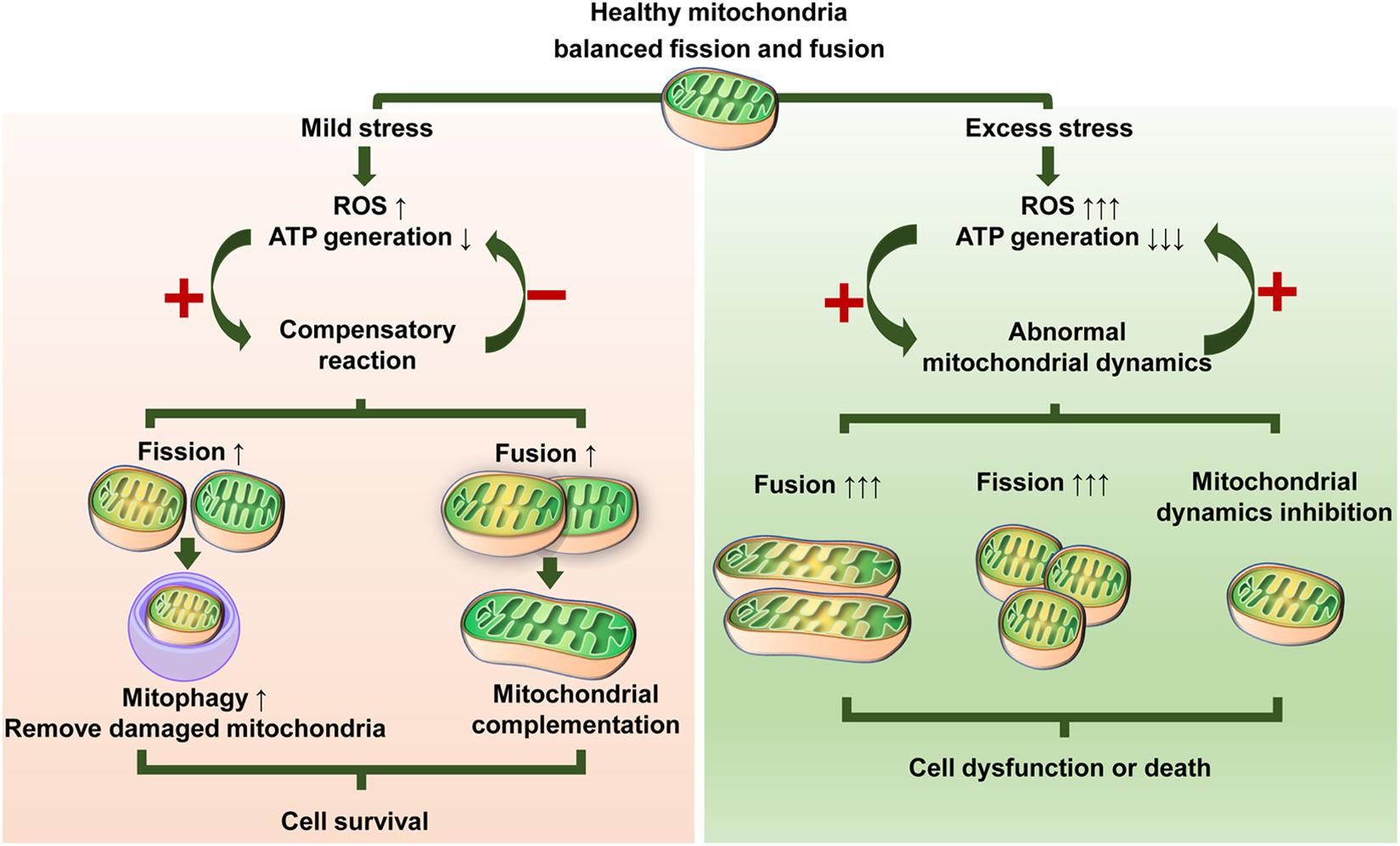
Emerging evidence suggests that mitochondrial dynamics are key contributors to stem cell fate determination. MSCs undergo specific mitochondrial dynamics during proliferation, migration, differentiation, apoptosis, or aging. Mitochondria are highly dynamic organelles that maintain their morphology via continuous fission and fusion, also known as mitochondrial dynamics. Mesenchymal stem cells (MSCs) are pivotal to tissue homeostasis, repair, and regeneration due to their potential for self-renewal, multilineage differentiation, and immune modulation.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
